pipeline
COVID-19
| Optimization | Pre-clinical | ADMET/CMC | Ph Ⅰ | Ph Ⅱ |
|---|---|---|---|---|
|
IV
|
||||
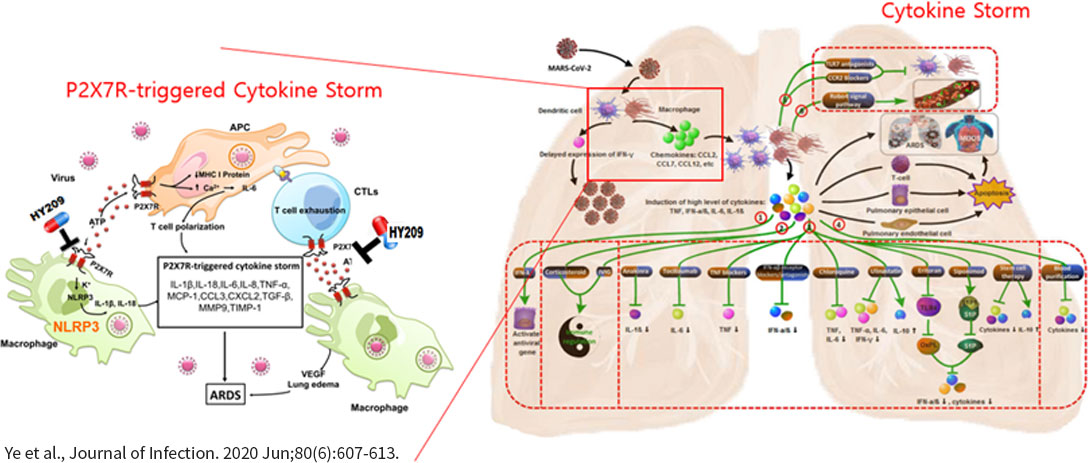
NuSepin® is IV injection form of HY209 for the treatment of systemic inflammatory response syndrome. Safety and tolerability of IV administration is confirmed through phase 1 study. Phase 2 study for COVID-19 pneumonia was successfully done in Europe with 64 patients of uncontrolled inflammatory responses after the SARS-CoV-2 virus infection.
In both LPS- and CLP-induced septic mouse model, NuSepin® treatment strongly suppressed IL-1β, IL-6, TNF-α, and MCP-1, pro-inflammatory cytokines associated with cytokine storms, improved the functions of liver and kidney which were damaged by cytokine storms, leading to the survival rate of 70%~90% depending on animal models. Similar efficacies were observed in a separate septic canine model.
Safety and tolerability study (phase 1) was completed with no reported SAEs, leading to a randomized, double blinded, placebo-controlled phase 2 study for COVID-19 patients conducted at 5 clinical sites in Romania. Excellent clinical improvement for hospitalized COVID-19 patients was demonstrated in phase 2a study. Multinational phase 2b/3 study for COVID-19 pneumonia is ongoing with KDDF (Korea Drug Development Fund) sponsorship.