pipeline
Atopic Dermatitis
| Optimization | Pre-clinical | ADMET/CMC | Ph Ⅰ | Ph Ⅱ |
|---|---|---|---|---|
|
TO
|
||||
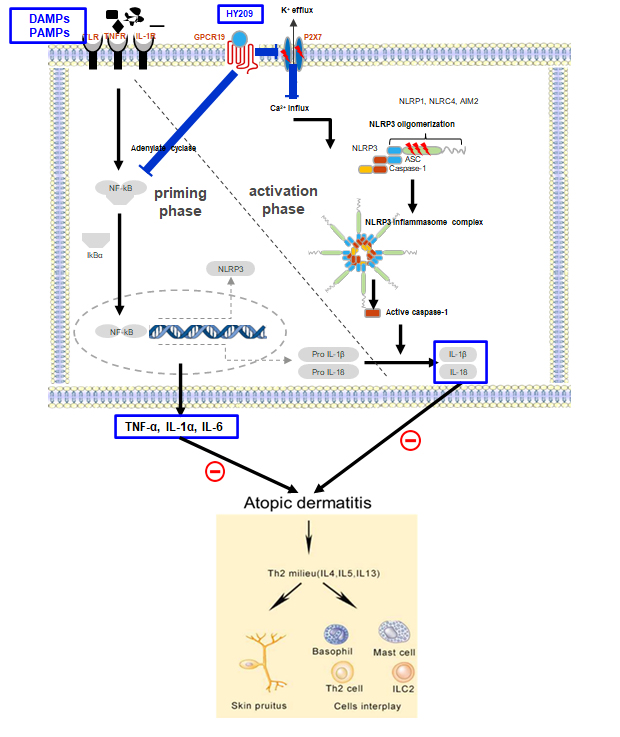
NuGel® is a gel type atopic dermatitis drug candidate, inhibiting NLRP3 inflammasome through GPCR19-P2X7 modulation. Its active pharmaceutical ingredient called HY209 has demonstrated a strong efficacy in atopic mouse model, suppressing a broad spectrum of pro-inflammatory cytokines such as TNF-α, IL-1β, IL-4, IL-6, IL-13, IL-17, MCP-1(CCL2), RANTES, etc. Currently, phase 2 clinical study is on-going in Korea.
In DNCB atopic mouse model, NuGel has shown more rapid tissue regeneration effects than current standard of care treatment such as corticosteroid, calcineurin inhibitor as well as new treatments such as PDE4 inhibitor and JAK inhibitor.
NuGel works on both innate immunity and adaptive immunity by significantly reducing the number of infiltrating immune cells (mast cells, eosinophils, MPOs) while increasing the number of regulatory T cells and upregulating the expression of IL-10.
Safety and tolerability study (phase 1) has been completed with no reported SAEs or systemic absorption, Safety, tolerability and efficacy study (phase 1 & 2) have been completed with excellent efficacy and safety profile, preparing for phase 2 study in US.